Iron is a chemical element with symbol Fe (from Latin:ferrum) and atomic number 26. It is a metal in the first transition series. It is by mass the most common element on Earth, forming much of Earth's outer and inner core.
It belongs to group 8 of the periodic table having trivial name null. You can also download Printable Periodic Table of Elements Flashcards for Iron in a PDF format.
Iron Facts
Read key information and facts about element Iron
| Name | Iron |
| Atomic Number | 26 |
| Atomic Symbol | Fe |
| Atomic Weight | 55.845 |
| Phase | Solid |
| Color | Gray |
| Appearance | lustrous metallic with a grayish tinge |
| Classification | Transition Metal |
| Natural Occurance | Primordial |
| Group in Periodic Table | 8 |
| Group Name | iron family |
| Period in Periodic Table | period 4 |
| Block in Periodic Table | d-block |
| Electronic Configuration | [Ar] 3d6 4s2 |
| Electronic Shell Structure (Electrons per shell) | 2, 8, 14, 2 |
| Melting Point | 1811 K |
| Boiling Point | 3134 K |
| CAS Number | CAS7439-89-6 |
How to Locate Iron on Periodic Table
Periodic table is arranged by atomic number, number of protons in the nucleus which is same as number of electrons. The atomic number increases from left to right. Periodic table starts at top left ( Atomic number 1) and ends at bottom right (atomic number 118). Therefore you can directly look for atomic number 26 to find Iron on periodic table.
Another way to read periodic table and locate an element is by using group number (column) and period number (row). To locate Iron on periodic table look for cross section of group 8 and period 4 in the modern periodic table.
Iron History
The element Iron was discovered by Unknown in year Before 5000 BCE . Iron was first isolated by Egypt in 4000 BCE. Iron derived its name from English word (ferrum in Latin).
| Discovered By | Unknown |
| Discovery Date | Before 5000 BCE |
| First Isolation | |
| Isolated by |
There is evidence that iron was known from before 5000 BCE. The oldest known iron objects used by humans are some beads of meteoric iron , made in Egypt in about 4000 BCE. The discovery of smelting around 3000 BCE led to the start of the iron age around 1200 BCE and the prominent use of iron for tools and weapons.
Iron Uses
Iron's main use is in making steel. When steel is combined with chromium, it produces stainless steel, which is resistant to corrosion
Iron Presence: Abundance in Nature and Around Us
The table below shows the abundance of Iron in Universe, Sun, Meteorites, Earth's Crust, Oceans and Human Body.
| ppb by weight (1ppb =10^-7 %) | ppb by atoms (1ppb =10^-7 %) | |
|---|---|---|
| Abundance in Universe | 1100000 | 20000 |
| Abundance in Sun | 1000000 | 30000 |
| Abundance in Meteorites | 220000000 | 77000000 |
| Abundance in Earth's Crust | 63000000 | 23000000 |
| Abundance in Oceans | 3 | 0.33 |
| Abundance in Humans | 60000 | 6700 |
Crystal Structure of Iron
The solid state structure of Iron is Body Centered Cubic.
The Crystal structure can be described in terms of its unit Cell. The unit Cells repeats itself in three dimensional space to form the structure.
Unit Cell Parameters
The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges Lattice Constants (a, b and c)
| a | b | c |
|---|---|---|
| 286.65 pm | 286.65 pm | 286.65 pm |
and the angles between them Lattice Angles (alpha, beta and gamma).
| alpha | beta | gamma |
|---|---|---|
| π/2 | π/2 | π/2 |
The positions of the atoms inside the unit cell are described by the set of atomic positions ( xi, yi, zi) measured from a reference lattice point.
The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in three-dimensional space are described by the 230 space groups (219 distinct types, or 230 if chiral copies are considered distinct.
| Space Group Name | Im_ 3m |
| Space Group Number | 229 |
| Crystal Structure | Body Centered Cubic |
| Number of atoms per unit cell | 2 |
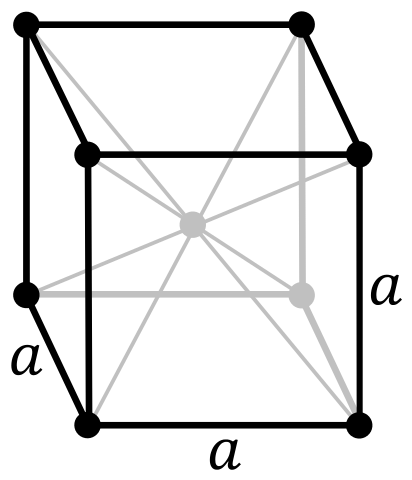
The number of atoms per unit cell in a simple cubic, face-centered cubic and body-centred cubic are 1,4,2 respectively.
Iron Atomic and Orbital Properties
Iron atoms have 26 electrons and the electronic shell structure is [2, 8, 14, 2] with Atomic Term Symbol (Quantum Numbers) 5D4.
| Atomic Number | 26 |
| Number of Electrons (with no charge) | 26 |
| Number of Protons | 26 |
| Mass Number | 56 |
| Number of Neutrons | 30 |
| Shell structure (Electrons per energy level) | 2, 8, 14, 2 |
| Electron Configuration | [Ar] 3d6 4s2 |
| Valence Electrons | 3d6 4s2 |
| Valence (Valency) | 3 |
| Main Oxidation States | 2, 3 |
| Oxidation States | -4, -2, -1, 0, 1, 2, 3, 4, 5, 6, 7 |
| Atomic Term Symbol (Quantum Numbers) | 5D4 |
Bohr Atomic Model of Iron - Electrons per energy level
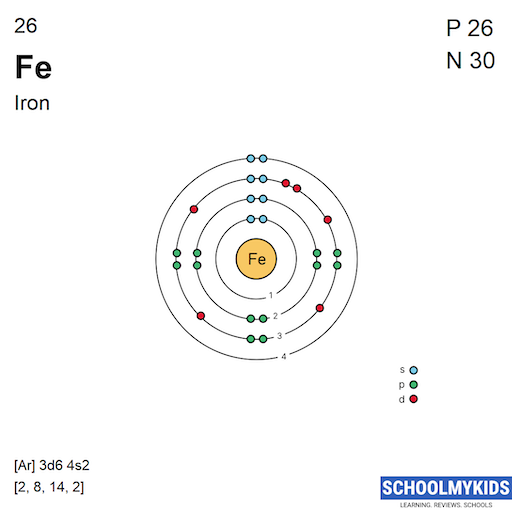
| n | s | p | d | f |
|---|
Ground State Electronic Configuration of Iron - neutral Iron atom
Abbreviated electronic configuration of Iron
The ground state abbreviated electronic configuration of Neutral Iron atom is [Ar] 3d6 4s2. The portion of Iron configuration that is equivalent to the noble gas of the preceding period, is abbreviated as [Ar]. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used. This is important as it is the Valence electrons 3d6 4s2, electrons in the outermost shell that determine the chemical properties of the element.
Unabbreviated electronic configuration of neutral Iron
Complete ground state electronic configuration for the Iron atom, Unabbreviated electronic configuration
1s2 2s2 2p6 3s2 3p6 3d6 4s2
Electrons are filled in atomic orbitals as per the order determined by the Aufbau principle, Pauli Exclusion Principle and Hund’s Rule.
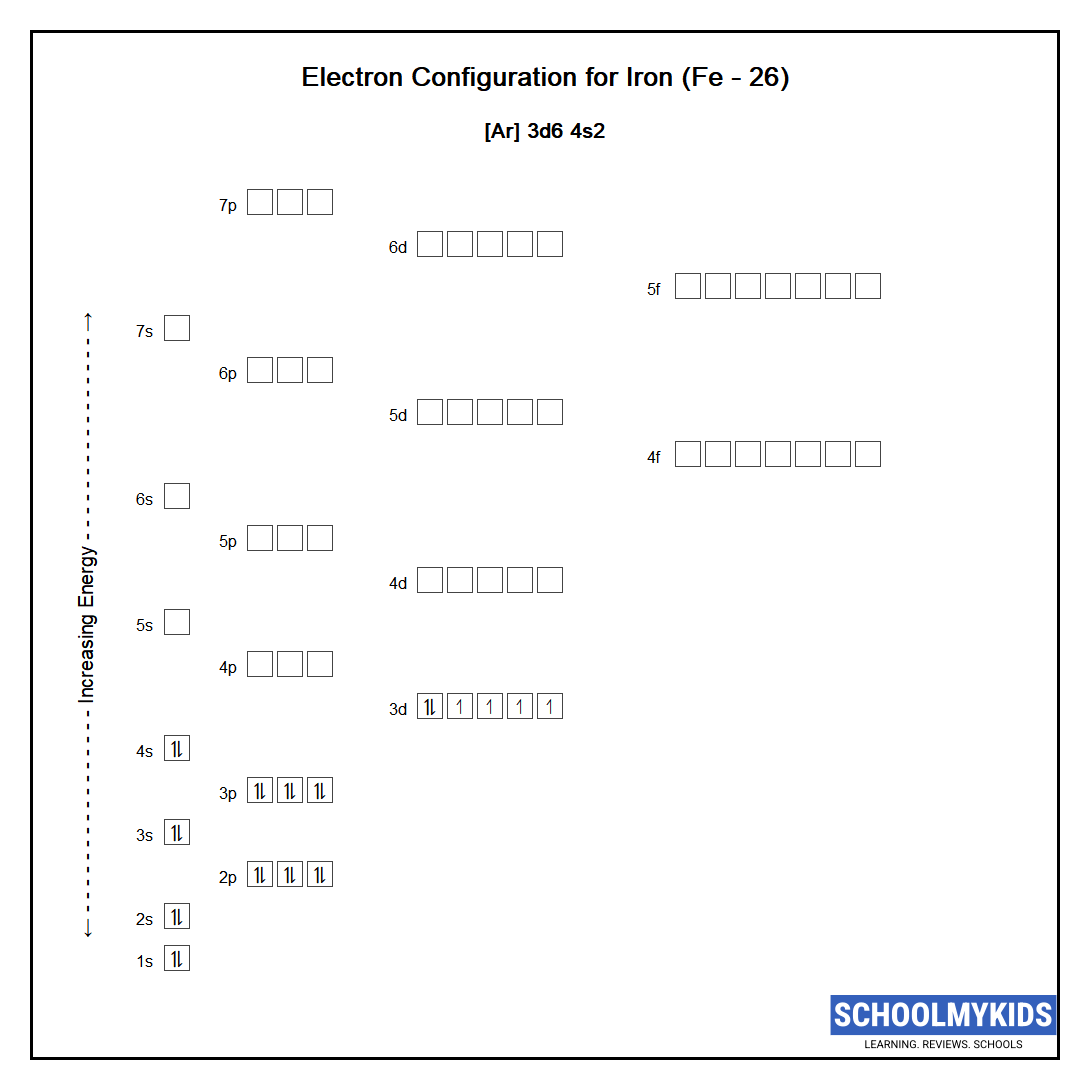
Atomic Structure of Iron
Iron atomic radius is 156 pm, while it's covalent radius is 125 pm.
| Atomic Radius Calculated | 156 pm(1.56 Å) |
| Atomic Radius Empirical | 140 pm (1.4 Å) |
| Atomic Volume | 7.0923 cm3/mol |
| Covalent Radius | 125 pm (1.25 Å) |
| Van der Waals Radius | - |
| Neutron Cross Section | 2.56 |
| Neutron Mass Absorption | 0.0015 |
Spectral Lines of Iron - Atomic Spectrum of Iron
A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from an excess or deficiency of photons in a narrow frequency range, compared with the nearby frequencies. Spectral lines are often used to identify atoms and molecules.
Spectral lines are the result of interaction between a quantum system and a single photon. A spectral line may be observed either as an emission line or an absorption line.
Spectral lines are highly atom-specific, and can be used to identify the chemical composition of any medium. Several elements, including helium, thallium, and caesium, were discovered by spectroscopic means. They are widely used to determine the physical conditions of stars and other celestial bodies that cannot be analyzed by other means.
Emission spectrum of Iron
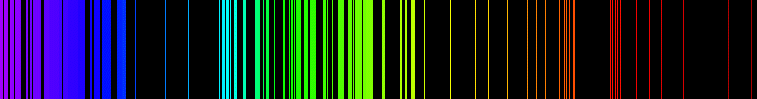
Absorption spectrum of Iron

Iron Chemical Properties: Iron Ionization Energies and electron affinity
The electron affinity of Iron is 15.7 kJ/mol.
| Valence | 3 |
| Electronegativity | 1.83 |
| ElectronAffinity | 15.7 kJ/mol |
Ionization Energy of Iron
Ionization energy is the amount of energy required to remove an electron from an atom or molecule.in chemistry, this energy is expresed in kilocalories per mole (kcal/mol) or kilojoules per mole (kJ/mol).
Refer to table below for Ionization energies of Iron
| Ionization energy number | Enthalpy - kJ/mol |
|---|---|
| 1st | 762.5 |
| 2nd | 1561.9 |
| 3rd | 2957 |
| 4th | 5290 |
| 5th | 7240 |
| 6th | 9560 |
| 7th | 12060 |
| 8th | 14580 |
| 9th | 22540 |
| 10th | 25290 |
| 11th | 28000 |
| 12th | 31920 |
| 13th | 34830 |
| 14th | 37840 |
| 15th | 44100 |
| 16th | 47206 |
| 17th | 122200 |
| 18th | 131000 |
| 19th | 140500 |
| 20th | 152600 |
| 21st | 163000 |
| 22nd | 173600 |
| 23rd | 188100 |
| 24th | 195200 |
| 25th | 851800 |
| 26th | 895161 |
Iron Physical Properties
Refer to below table for Iron Physical Properties
| Density | 7.874 g/cm3(when liquid at m.p density is $6.98 g/cm3) |
| Molar Volume | 7.0923 cm3/mol |
Elastic Properties
| Young Modulus | 211 |
| Shear Modulus | 82 GPa |
| Bulk Modulus | 170 GPa |
| Poisson Ratio | 0.29 |
Hardness of Iron - Tests to Measure of Hardness of Element
| Mohs Hardness | 4 MPa |
| Vickers Hardness | 608 MPa |
| Brinell Hardness | 490 MPa |
Iron Electrical Properties
Electrical resistivity measures element's electrical resistance or how strongly it resists electric current.The SI unit of electrical resistivity is the ohm-metre (Ω⋅m). While Electrical conductivity is the reciprocal of electrical resistivity. It represents a element's ability to conduct electric current. The SI unit of electrical conductivity is siemens per metre (S/m).
Iron is a conductor of electricity. Refer to table below for the Electrical properties of Iron
| Electrical conductors | Conductor |
| Electrical Conductivity | 10000000 S/m |
| Resistivity | 9.7e-8 m Ω |
| Superconducting Point | - |
Iron Heat and Conduction Properties
| Thermal Conductivity | 80 W/(m K) |
| Thermal Expansion | 0.0000118 /K |
Iron Magnetic Properties
| Magnetic Type | Ferromagnetic |
| Curie Point | 1043 K |
| Mass Magnetic Susceptibility | - |
| Molar Magnetic Susceptibility | - |
| Volume Magnetic Susceptibility | - |
Optical Properties of Iron
| Refractive Index | - |
Acoustic Properties of Iron
| Speed of Sound | 4910 m/s |
Iron Thermal Properties - Enthalpies and thermodynamics
Refer to table below for Thermal properties of Iron
| Melting Point | 1811 K(1537.85 °C, 2800.130 °F) |
| Boiling Point | 3134 K(2860.85 °C, 5181.530 °F) |
| Critical Temperature | - |
| Superconducting Point | - |
Enthalpies of Iron
| Heat of Fusion | 13.8 kJ/mol |
| Heat of Vaporization | 347 kJ/mol |
| Heat of Combustion | - |
Iron Isotopes - Nuclear Properties of Iron
Iron has 28 isotopes, with between 45 and 72 nucleons. Iron has 4 stable naturally occuring isotopes.
Isotopes of Iron - Naturally occurring stable Isotopes: 54Fe, 56Fe, 57Fe, 58Fe.
| Isotope | Z | N | Isotope Mass | % Abundance | T half | Decay Mode |
|---|---|---|---|---|---|---|
| 45Fe | 26 | 19 | 45 | Synthetic | ||
| 46Fe | 26 | 20 | 46 | Synthetic | ||
| 47Fe | 26 | 21 | 47 | Synthetic | ||
| 48Fe | 26 | 22 | 48 | Synthetic | ||
| 49Fe | 26 | 23 | 49 | Synthetic | ||
| 50Fe | 26 | 24 | 50 | Synthetic | ||
| 51Fe | 26 | 25 | 51 | Synthetic | ||
| 52Fe | 26 | 26 | 52 | Synthetic | ||
| 53Fe | 26 | 27 | 53 | Synthetic | ||
| 54Fe | 26 | 28 | 54 | 5.845% | Stable | N/A |
| 55Fe | 26 | 29 | 55 | Synthetic | ||
| 56Fe | 26 | 30 | 56 | 91.754% | Stable | |
| 57Fe | 26 | 31 | 57 | 2.119% | Stable | N/A |
| 58Fe | 26 | 32 | 58 | 0.282% | Stable | N/A |
| 59Fe | 26 | 33 | 59 | Synthetic | ||
| 60Fe | 26 | 34 | 60 | Synthetic | ||
| 61Fe | 26 | 35 | 61 | Synthetic | ||
| 62Fe | 26 | 36 | 62 | Synthetic | ||
| 63Fe | 26 | 37 | 63 | Synthetic | ||
| 64Fe | 26 | 38 | 64 | Synthetic | ||
| 65Fe | 26 | 39 | 65 | Synthetic | ||
| 66Fe | 26 | 40 | 66 | Synthetic | ||
| 67Fe | 26 | 41 | 67 | Synthetic | ||
| 68Fe | 26 | 42 | 68 | Synthetic | ||
| 69Fe | 26 | 43 | 69 | Synthetic | ||
| 70Fe | 26 | 44 | 70 | Synthetic | ||
| 71Fe | 26 | 45 | 71 | Synthetic | ||
| 72Fe | 26 | 46 | 72 | Synthetic |
Regulatory and Health - Health and Safety Parameters and Guidelines
The United States Department of Transportation (DOT) identifies hazard class of all dangerous elements/goods/commodities either by its class (or division) number or name. The DOT has divided these materials into nine different categories, known as Hazard Classes.
| DOT Numbers | 3089 |
| DOT Hazard Class | 4.1 |
NFPA 704 is a Standard System for the Identification of the Hazards of Materials for Emergency Response. NFPA is a standard maintained by the US based National Fire Protection Association.
The health (blue), flammability (red), and reactivity (yellow) rating all use a numbering scale ranging from 0 to 4. A value of zero means that the element poses no hazard; a rating of four indicates extreme danger.
| NFPA Fire Rating | 1 | Flash Points Above 93.3°C (200°F) |
| NFPA Health Rating | 1 | Flash Points Above 93.3°C (200°F) |
| NFPA Reactivity Rating | 0 | Will not burn |
| NFPA Hazards |
| Autoignition Point | 100 °C |
| Flashpoint | - |
Database Search
List of unique identifiers to search the element in various chemical registry databases
| Database | Identifier number |
|---|---|
| CAS Number - Chemical Abstracts Service (CAS) | CAS7439-89-6 |
| RTECS Number | RTECSNO4565500 |
| CID Number | CID23925 |
| Gmelin Number | - |
| NSC Number | - |
Compare Iron with other elements
Compare Iron with all Group 8 elements
Compare Iron with all Period 4 elements
Compare Iron with all Transition Metal elements
FAQs
What is the electronic configuration of Iron?
The electronic configuration of Iron is 1s2 2s2 2p6 3s2 3p6 3d6 4s2.
What is the abbreviated electronic configuration of Iron?
The abbreviated electronic configuration of Iron is [Ar] 3d6 4s2. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets.
What is the symbol of Iron?
Symbol of Iron is Fe. Iron is a chemical element with symbol Fe and atomic number 26.
What is the position of Iron in the Periodic Table?
Iron is a chemical element with the symbol Fe and atomic number 26. Iron is the 26 element on the periodic table. It is located in group 8 and period 4 in the modern periodic table.
What is the atomic number of Iron?
The atomic number of Iron is 26.
What is the color of Iron?
Iron is of Gray color.
Who discovered Iron?
The element Iron was discovered by Unknown in year Before 5000 BCE . Iron was first isolated by Egypt in 4000 BCE.
How many valence electrons does a Iron atom have?
Iron has 3 valence electrons. Iron has 26 electrons out of which 3 valence electrons are present in the 3d6 4s2 outer orbitals of atom.
What is the melting Point of Iron?
Melting Point of Iron is 1811 K.
What is the boiling Point of Iron?
Boiling Point of Iron is 3134 K.
What is the melting Point of Iron in Kelvin?
Melting Point of Iron in Kelvin is 1811 K.
What is the boiling Point of Iron in Kelvin?
Boiling Point of Iron in Kelvin is 3134 K.
What is the melting Point of Iron in Celsius?
Melting Point of Iron in Celsius is 1537.85 °C.
What is the boiling Point of Iron in Celsius?
Boiling Point of Iron in Celsius is 2860.85 °C.
What is the melting Point of Iron in Fahrenheit?
Melting Point of Iron in Fahrenheit is 2800.13 °F.
What is the boiling Point of Iron in Fahrenheit?
Boiling Point of Iron in Fahrenheit is 5181.53 °F.
What is the electronic configuration of Iron 26?
The electronic configuration of Iron will be 1s2 2s2 2p6 3s2 3p6 3d6 4s2.
How do you write the electron configuration for Iron?
The electronic configuration of Iron will be 1s2 2s2 2p6 3s2 3p6 3d6 4s2.

