Periodic Table Element Comparison: Compare Elements - Sulfur vs Antimony
Compare Sulfur and Antimony
Compare Sulfur and Antimony on the basis of their properties, attributes and periodic table facts. Compare elements on more than 90 properties. All the elements of similar categories show a lot of similarities and differences in their chemical, atomic, physical properties and uses. These similarities and dissimilarities should be known while we study periodic table elements. You can study the detailed comparison between Sulfur vs Antimony with most reliable information about their properties, attributes, facts, uses etc. You can compare S vs Sb on more than 90 properties like electronegativity , oxidation state, atomic shells, orbital structure, Electronaffinity, physical states, electrical conductivity and many more.
Facts
| Name | Sulfur | Antimony |
| Atomic Number | 16 | 51 |
| Atomic Symbol | S | Sb |
| Atomic Weight | 32.065 | 121.76 |
| Phase at STP | Solid | Solid |
| Color | Yellow | Silver |
| Metallic Classification | Other Nonmetal | Metalloid |
| Group in Periodic Table | group 16 | group 15 |
| Group Name | oxygen family | nitrogen family |
| Period in Periodic Table | period 3 | period 5 |
| Block in Periodic Table | p -block | p -block |
| Electronic Configuration | [Ne] 3s2 3p4 | [Kr] 4d10 5s2 5p3 |
| Electronic Shell Structure (Electrons per shell) | 2, 8, 6 | 2, 8, 18, 18, 5 |
| Melting Point | 388.36 K | 903.78 K |
| Boiling Point | 717.87 K | 1860 K |
| CAS Number | CAS7704-34-9 | CAS7440-36-0 |
| Neighborhood Elements | Neighborhood Elements of Sulfur | Neighborhood Elements of Antimony |
History
| Name | Sulfur | Antimony |
| History | The element Sulfur was discovered by Chinese/Indians in year Before 2000 BCE. Sulfur derived its name from the Latin word sulphur, 'fire and brimstone'. | The element Antimony was discovered by Arabic alchemist in year ca. 800 AD in one. Antimony derived its name from the Greek anti, 'against', and monos, 'alone' (stibium in Latin). |
| Discovery | Chinese/Indians (Before 2000 BCE) | Arabic alchemist (ca. 800 AD) |
| Isolated | () | () |
Presence: Abundance in Nature and Around Us
Parts per billion (ppb) by weight / by atoms (1ppb =10^-7 %)
| Name | Sulfur | Antimony |
| Abundance in Universe | 500000 / 20000 | 0.4 / 0.004 |
| Abundance in Sun | 400000 / 10000 | 1 / 0.01 |
| Abundance in Meteorites | 41000000 / 22000000 | 120 / 20 |
| Abundance in Earth's Crust | 420000 / 270000 | 200 / 30 |
| Abundance in Oceans | 928000 / 179000 | 0.2 / 0.010 |
| Abundance in Humans | 2000000 / 390000 | - / - |
Crystal Structure and Atomic Structure
| Name | Sulfur | Antimony |
| Atomic Volume | 15.53 cm3/mol | 18.181 cm3/mol |
| Atomic Radius | 88 pm | 133 pm |
| Covalent Radius | 102 pm | 138 pm |
| Van der Waals Radius | 180 pm | 206 pm |
| Atomic Spectrum - Spectral Lines | ||
| Emission Spectrum |  |  |
| Absorption Spectrum |  |  |
| Lattice Constant | 1043.7, 1284.5, 2436.9 pm | 430.7, 430.7, 1127.3 pm |
| Lattice Angle | π/2, π/2, π/2 | π/2, π/2, 2 π/3 |
| Space Group Name | Fddd | R_ 3m |
| Space Group Number | 70 | 166 |
| Crystal Structure | Face Centered Orthorhombic 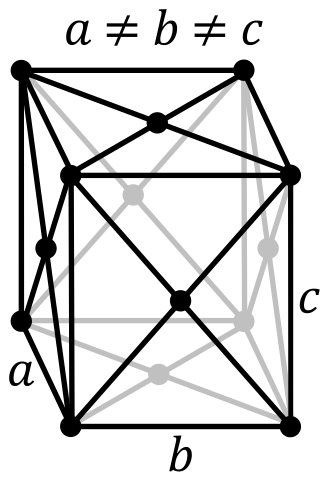 | Simple Trigonal 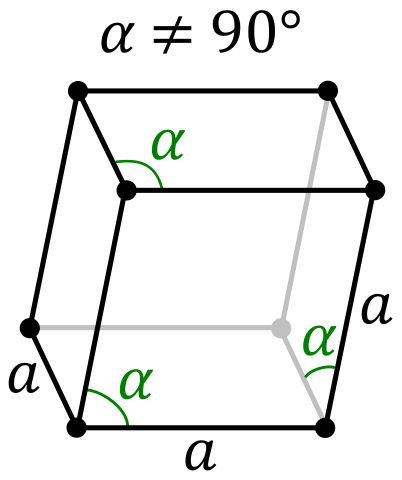 |
Atomic and Orbital Properties
| Name | Sulfur | Antimony |
| Atomic Number | 16 | 51 |
| Number of Electrons (with no charge) | 16 | 51 |
| Number of Protons | 16 | 51 |
| Mass Number | 32.065 | 121.76 |
| Number of Neutrons | 16 | 71 |
| Shell structure (Electrons per energy level) | 2, 8, 6 | 2, 8, 18, 18, 5 |
| Electron Configuration | [Ne] 3s2 3p4 | [Kr] 4d10 5s2 5p3 |
| Valence Electrons | 3s2 3p4 | 5s2 5p3 |
| Oxidation State | -2, 2, 4, 6 | -3, 3, 5 |
| Atomic Term Symbol (Quantum Numbers) | 3P2 | 4S3/2 |
| Shell structure | 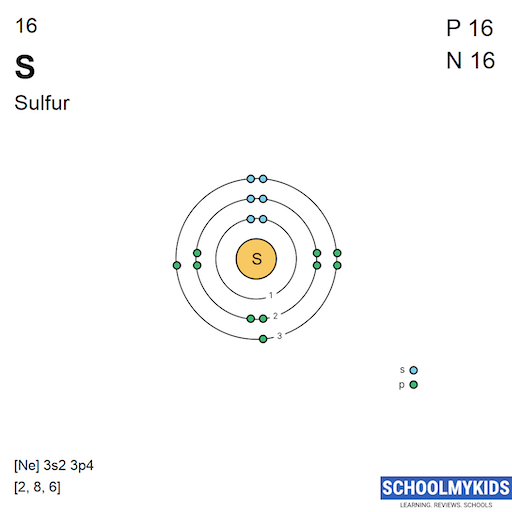 | 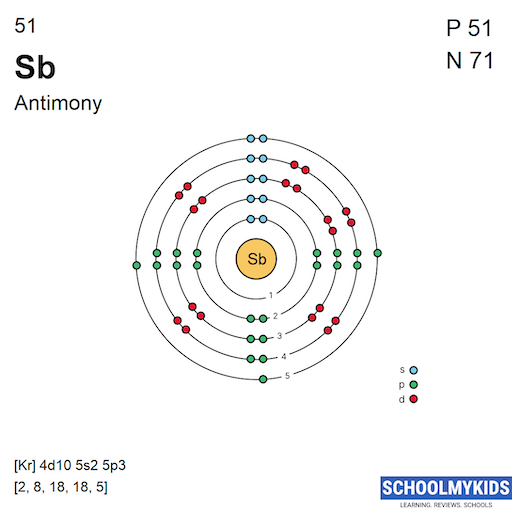 |
Isotopes and Nuclear Properties
Sulfur has 4 stable naturally occuring isotopes while Antimony has 2 stable naturally occuring isotopes.
| Name | Sulfur | Antimony |
| Known Isotopes | 26S, 27S, 28S, 29S, 30S, 31S, 32S, 33S, 34S, 35S, 36S, 37S, 38S, 39S, 40S, 41S, 42S, 43S, 44S, 45S, 46S, 47S, 48S, 49S | 103Sb, 104Sb, 105Sb, 106Sb, 107Sb, 108Sb, 109Sb, 110Sb, 111Sb, 112Sb, 113Sb, 114Sb, 115Sb, 116Sb, 117Sb, 118Sb, 119Sb, 120Sb, 121Sb, 122Sb, 123Sb, 124Sb, 125Sb, 126Sb, 127Sb, 128Sb, 129Sb, 130Sb, 131Sb, 132Sb, 133Sb, 134Sb, 135Sb, 136Sb, 137Sb, 138Sb, 139Sb |
| Stable Isotopes | Naturally occurring stable isotopes: 32S, 33S, 34S, 36S | Naturally occurring stable isotopes: 121Sb, 123Sb |
| Neutron Cross Section | 0.52 | 5.4 |
| Neutron Mass Absorption | 0.00055 | 0.0016 |
Chemical Properties: Ionization Energies and electron affinity
| Name | Sulfur | Antimony |
| Valence or Valency | 6 | 5 |
| Electronegativity | 2.58 Pauling Scale | 2.05 Pauling Scale |
| Electron Affinity | 200 kJ/mol | 103.2 kJ/mol |
| Ionization Energies | 1st: 999.6 kJ/mol 2nd: 2252 kJ/mol 3rd: 3357 kJ/mol 4th: 4556 kJ/mol 5th: 7004.3 kJ/mol 6th: 8495.8 kJ/mol 7th: 27107 kJ/mol 8th: 31719 kJ/mol 9th: 36621 kJ/mol 10th: 43177 kJ/mol 11th: 48710 kJ/mol 12th: 54460 kJ/mol 13th: 62930 kJ/mol 14th: 68216 kJ/mol 15th: 311048 kJ/mol 16th: 337138 kJ/mol | 1st: 834 kJ/mol 2nd: 1594.9 kJ/mol 3rd: 2440 kJ/mol 4th: 4260 kJ/mol 5th: 5400 kJ/mol 6th: 10400 kJ/mol |
Physical Properties
| Name | Sulfur | Antimony |
| Density | 1.96 g/cm3 | 6.697 g/cm3 |
| Molar Volume | 15.53 cm3/mol | 18.181 cm3/mol |
Elastic Properties | ||
| Young Modulus | - | 55 |
| Shear Modulus | - | 20 GPa |
| Bulk Modulus | 7.7 GPa | 42 GPa |
| Poisson Ratio | - | - |
Hardness - Tests to Measure of Hardness of Element | ||
| Mohs Hardness | 2 MPa | 3 MPa |
| Vickers Hardness | - | - |
| Brinell Hardness | - | 294 MPa |
Electrical Properties | ||
| Electrical Conductivity | 1e-15 S/m | 2500000 S/m |
| Resistivity | 1000000000000000 m Ω | 4.17e-7 m Ω |
| Superconducting Point | - | - |
Heat and Conduction Properties | ||
| Thermal Conductivity | 0.205 W/(m K) | 24 W/(m K) |
| Thermal Expansion | - | 0.000011 /K |
Magnetic Properties | ||
| Magnetic Type | Diamagnetic | Diamagnetic |
| Curie Point | - | - |
| Mass Magnetic Susceptibility | -6.2e-9 m3/kg | -1.09e-8 m3/kg |
| Molar Magnetic Susceptibility | -1.99e-10 m3/mol | -1.327e-9 m3/mol |
| Volume Magnetic Susceptibility | -0.0000122 | -0.000073 |
Optical Properties | ||
| Refractive Index | 1.001111 | - |
Acoustic Properties | ||
| Speed of Sound | - | 3420 m/s |
Thermal Properties - Enthalpies and thermodynamics
| Name | Sulfur | Antimony |
| Melting Point | 388.36 K | 903.78 K |
| Boiling Point | 717.87 K | 1860 K |
| Critical Temperature | 1314 K | - |
| Superconducting Point | - | - |
Enthalpies | ||
| Heat of Fusion | 1.73 kJ/mol | 19.7 kJ/mol |
| Heat of Vaporization | 9.8 kJ/mol | 68 kJ/mol |
| Heat of Combustion | - | - |
Regulatory and Health - Health and Safety Parameters and Guidelines
| Name | Sulfur | Antimony |
| CAS Number | CAS7704-34-9 | CAS7440-36-0 |
| RTECS Number | {RTECSWS4250000, N/A, N/A, N/A, N/A, N/A} | {RTECSCC4025000} |
| DOT Hazard Class | 4.1 | 6.1 |
| DOT Numbers | 1350 | 2871 |
| EU Number | - | - |
| NFPA Fire Rating | - | - |
| NFPA Health Rating | - | - |
| NFPA Reactivity Rating | - | - |
| NFPA Hazards | - | - |
| AutoIgnition Point | - | - |
| Flashpoint | - | - |
Compare With Other Elements
Compare Sulfur with all Group 16 elementsCompare Sulfur with all Period 3 elementsSulfur vs SodiumSulfur vs MagnesiumSulfur vs AluminiumSulfur vs SiliconSulfur vs PhosphorusSulfur vs ChlorineSulfur vs Argon Compare Sulfur with all Other Nonmetal elements | Compare Antimony with all Group 15 elementsAntimony vs NitrogenAntimony vs PhosphorusAntimony vs ArsenicAntimony vs BismuthAntimony vs Moscovium Compare Antimony with all Period 5 elementsAntimony vs RhodiumAntimony vs RubidiumAntimony vs StrontiumAntimony vs YttriumAntimony vs ZirconiumAntimony vs TinAntimony vs NiobiumAntimony vs MolybdenumAntimony vs TechnetiumAntimony vs RutheniumAntimony vs PalladiumAntimony vs SilverAntimony vs CadmiumAntimony vs IndiumAntimony vs TelluriumAntimony vs IodineAntimony vs Xenon Compare Antimony with all Metalloid elements |


